Nearly half of all Canadian adults have experienced tinnitus in their lifetime and a new at-home tinnitus treatment device offers chronic sufferers hope.
- Lenire tinnitus treatment device now available in Canada through 21 clinics across Alberta, British Colombia, Manitoba, Ontario, and Québec.
- 43% of Canadians (11.2m) report experiencing tinnitus in their lifetime with 37% of Canadians (9.2m) report experiencing tinnitus in the past year according to data from a Canadian Health Measures Survey (CHMS).1
- 84% of 517 tinnitus patients across two large-scale clinical trials reported relief from tinnitus that sustained for at least 12-months.2,3
- 91.5% of tinnitus patients reported significant relief from tinnitus in a peer-reviewed analysis of patients treated with Lenire at an independent clinic.4
“Lenire has been a miracle that has changed my life” – Toronto-based tinnitus patient, Jim Gray.
Toronto, Canada – Tuesday, April 7, 2026: Neuromod Devices, the medical technology company that specialises in tinnitus treatment technology, has made the Lenire tinnitus treatment device available in Canada.
Lenire will be exclusively available through hearing care clinics with expertise in tinnitus in Ontario, British Columbia, Alberta, Manitoba, and Québec. Lenire will be available in 21 clinics as part of the device’s initial Canadian launch. Neuromod has planned further expansion in 2026.
Lenire® is the first bimodal neuromodulation device that has been proven to provide long-term relief from tinnitus. This has been proven across multiple large-scale clinical trials. 2,3 The device combines mild pulses to the surface of the tongue via the ‘Tonguetip®’, with auditory stimulation. This dual stimulus drives changes to the brain and can deliver long-term relief from tinnitus.
Tinnitus is commonly known as ringing, hissing, or buzzing in the ears. 43% of Canadians report experiencing tinnitus in their lifetime. 37% report experiencing tinnitus in the past year according to a Canadian Health Measures Survey.1
“In the last few years, thousands of Canadians living with tinnitus have made contact with Neuromod to ask our team to make Lenire available in Canada. Today, I’m proud to say we have delivered for these people, the millions of others living with tinnitus, and for their healthcare providers,” said Neuromod Founder and CEO, Dr. Ross O’Neill. “A high prevalence of tinnitus in Canada is driving demand for clinically proven treatment options. Neuromod is excited to partner with leading tinnitus care clinics to make Lenire available to the millions of people living with constant noise in Canada.”
Lenire’s availability in Canada follows a comprehensive audit that verified Neuromod’s compliance with the international standard ISO13485 and regulatory requirements in multiple regulatory jurisdictions including Canada (Health Canada).
Clinical Trials Demonstrate Lenire Sustained Tinnitus Relief
Canada is the latest country to make Lenire® available in 2026, following successful launches in The Netherlands, Portugal, and further expansion in the USA.
Lenire is the first non-invasive bimodal neuromodulation tinnitus treatment device that has been proven effective in clinical trials and in real world patient analyses.
In 2023, the United States Food and Drug Administration (US FDA) granted De Novo approval to Lenire, making it the first and only bimodal neuromodulation device to achieve this milestone.
De Novo Approval from the FDA was granted based on the success of Lenire’s TENT-A3 controlled clinical trial. TENT-A3 demonstrated that Lenire was clinically superior for the treatment of tinnitus than sound-therapy. Sound-therapy was the trial’s control. 5 The Medical Device Single Audit Program (MDSAP) approved Lenire following the device’s FDA Approval. Lenire secured authorisation from Health Canada in late 2025.
Since then, Neuromod has been training hearing care professionals with tinnitus expertise throughout Canada to provide treatment with Lenire.
Nature – Scientific Reports published results from Lenire’s second large-scale clinical trial, TENT-A2. TENT-A2 showed 95% of compliant patients reported tinnitus improvement. 91% reported long-term relief that sustained for at least a year after treatment ended.1
“Hearing Solutions has been treating tinnitus patients for more than 25 years, and we are seeing more patients presenting to our clinicians with tinnitus as their primary concern, reflecting increased awareness and demand for care,” said Ron Abish, Founder, President and CEO of Hearing Solutions. “We are excited to expand the treatment options for our tinnitus patients with Lenire, a device that has demonstrated strong clinical trial results and real-world effectiveness.”
Watch: Toronto-based tinnitus patient share how Lenire gave him relief from tinnitus.
Lenire Proven Effective with Tinnitus Patients Treated at Independent Clinics
Results from Lenire’s real world patient analyses are consistent with the bimodal neuromodulation device’s clinical trial patient outcomes.
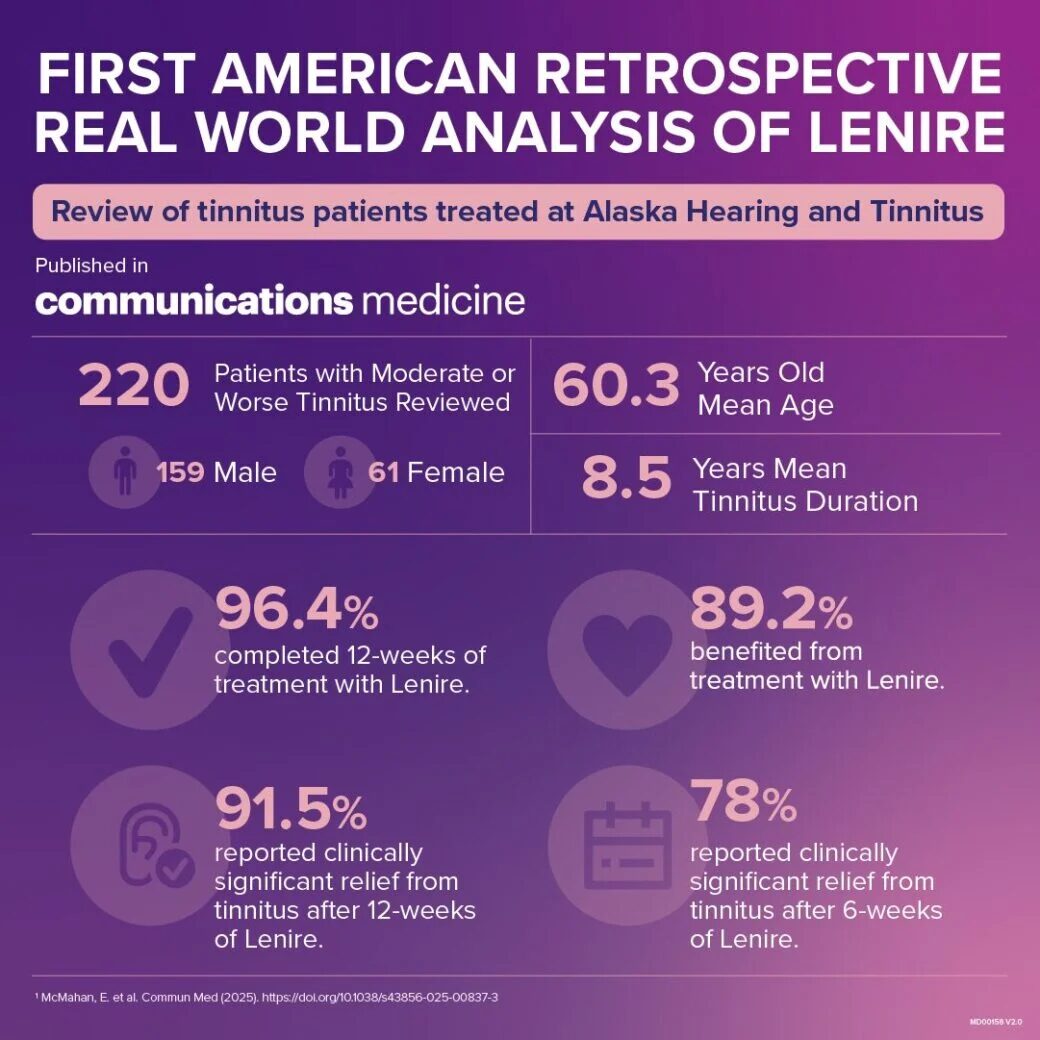
Nature Communications Medicine peer-reviewed and published the results of real world patients with bothersome tinnitus. The independent clinic, Alaska Hearing & Tinnitus, treated these patients with Lenire. 91.5% of 220 patients reported clinically meaningful reductions in tinnitus after treatment with Lenire.2
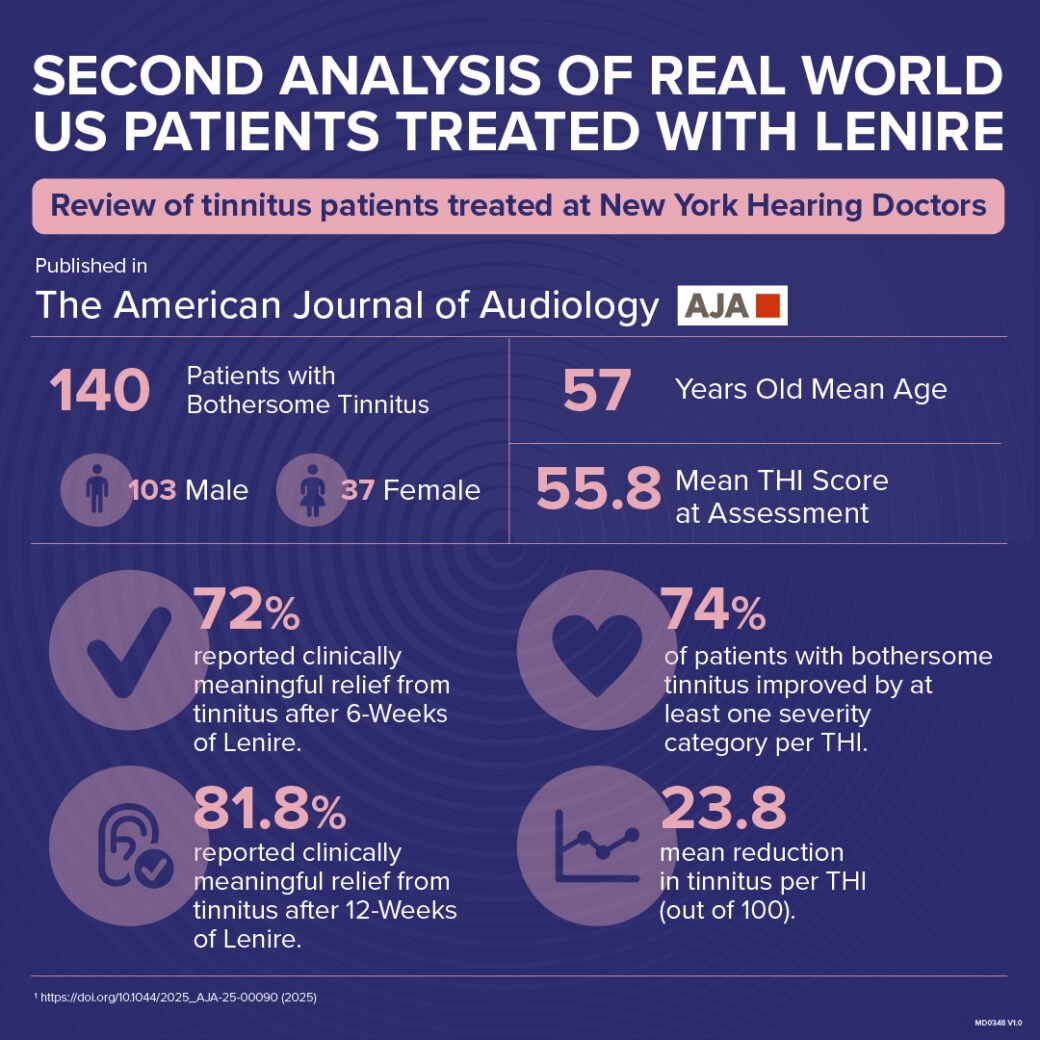
The American Journal of Audiology published results of 140 real world patients with bothersome tinnitus. The independent clinic, New York Hearing Doctors, treated these patients with Lenire. 81.8% of 140 patients reported clinically meaningful relief from tinnitus after treatment with Lenire.
“Due to my tinnitus, I had to take medication to get to sleep, which I hated to do and I was worried that it was going to get even worse.” said Toronto-based tinnitus patient, Jim Gray. “Lenire showed me that you can solve problems like tinnitus, it has been a miracle that has changed my life, “
Leading tinnitus charity, Tinnitus UK, rated Lenire as the top treatment option for safety and effectiveness. This rating acknowledges the large and ever-expanding evidence base for Lenire.
Lednire is available in Canada exclusively through hearing care clinics. These clinics are comprehensively trained to administer tinnitus care with Lenire following a suitability assessment. Patients can find a clinic providing Lenire in Canada by visiting www.lenire.com/find-a-clinic/.
About Neuromod Devices
Neuromod Devices is a global medical technology company with offices in Ireland and North America. The company specialises in the design and development of neuromodulation technologies to address the clinical needs of tinnitus patients.
Neuromod has completed extensive clinical trials. These trials aimed to confirm the safety and effectiveness of its non-invasive bimodal neuromodulation tinnitus treatment device, Lenire.
For more information about Neuromod visit www.neuromod.com.
References
- Ramage-Morin, P.L, Banks, R., Pineault, D., Atrach, M., Health Reports: Tinnitus in Canada, https://www.doi.org/10.25318/82-003-x201900300001-eng (2019)
- Conlon et al., Different bimodal neuromodulation settings reduce tinnitus symptoms in a large randomized trial, Sci Rep, nature.com/articles/s41598-022-13875-x (2022)
- Conlon et al., Sci. Transl. Med. 12, eabb2830 (2020)
- Mc Mahan, E., and Lim, H. Retrospective chart review demonstrating effectiveness of bimodal neuromodulation for tinnitus treatment, Commun Med (2025)
- Boedts, M., Beuchner, A., Khoo, S.G. et al Combining sound with tongue stimulation for the treatment of tinnitus. A multi-site single-arm controlled pivotal trial. Nature Comms. (2024)